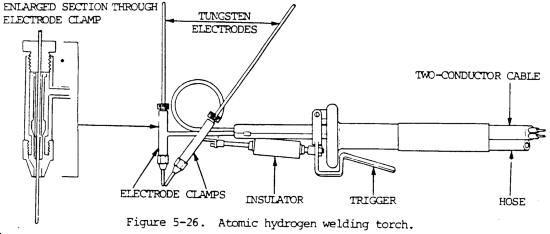
"A process in which the welding heat is generated by passing a stream of hydrogen through an electric arc between two inclined electrodes, which are usually of tungsten. The high temperature of the arc dissociates molecules of the gas into atoms, a large quantity of heat being absorbed by the hydrogen during dissociation. When the atoms leave the influence of the arc they recombine, forming molecules of hydrogen and liberating heat previously absorbed. The gas then burns in the ordinary way, taking up oxygen from the atmosphere for the purpose.
The average temperature of the flame is approximately 4000 deg. C., which is higher than the maximum temperature of any other flame. The heat is concentrated chiefly at the point of recombination of the atoms, and this recombination is accelerated catalytically by contact with the surface of the metal being welded. Thus an intense flame is obtained at the point of welding. The process is, therefore, used when rapid welding is necessary, as for stainless steels and other special alloys. The hydrogen envelope prevents oxidation both of the metal and the tungsten electrodes, and it also reduces the risk of nitrogen pick-up. The non-oxidizing characteristic is perhaps the most important in practice.
As a rule, the cost of welding by this process is slightly higher than with other processes, but it is sometimes the only practicable method by which a satisfactory weld can be made. An automatic atomic - hydrogen welding process has also been developed in which, instead of using hydrogen from high-pressure cylinders, the hydrogen is obtained by cracking anhydrous ammonia."
Odhams Practical & Technical Encyclopaedia 1947